Product #
Description
#
Add
F-35200
Phenolphthaleine presumptive test set for blood.
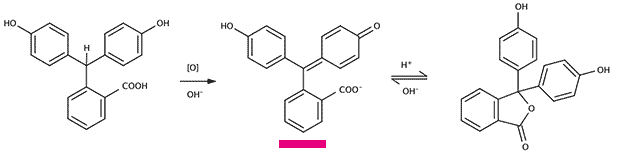
Left: phenolphthalin. Middle: after oxidation and removing of a proton by a base the pink phenolphthalein is formed. Right: in an acidic environment a colorless form of phenolphthalein is formed.
See also Wikipedia.








